NeuroServo announced FDA 510K clearance for VEEGix, its instant-use EEG
NeuroServo. is proud to announce that its innovative instant-use EEG has received U.S. Food and Drug Administration (FDA) clearance.
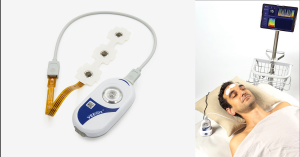
MONTREAL, QC, CANADA, August 26, 2024 /EINPresswire.com/ -- VEEGix is an instant-use, compact, prefrontal EEG device designed specifically for use in emergency situations within Intensive Care Units (ICUs) and Emergency Departments.Unlike traditional EEG systems, which typically require a specialized technician for setup and a neurologist for data interpretation, often resulting in a median lead time of around eight hours, VEEGix can be installed by nurses and provides a clear assessment of brain status within one minute.
VEEGix is not intended to replace full traditional EEG systems but is designed to serve two critical functions:
1. Emergency EEG Monitoring: In situations where time is of the essence and treatment windows can be less than an hour to prevent permanent brain damage, VEEGix provides rapid, reliable data.
2. Cerebral Monitoring for Patients in Intensive Care Units: VEEGix offers continuous monitoring for patients under sedation, helping healthcare providers track brain function with ease.
Following its clearance by Health Canada, VEEGix has now received FDA 510(k) approval, marking a significant milestone in the device’s journey to becoming a standard tool in medical practice.
“Each of the five vital signs became essential the day a simple monitoring device was made available,” said Nicolas Tremblay, CEO of NeuroServo. “With VEEGix, we are pioneering cerebral monitoring as the new vital sign by offering a device that can be operated and understood by any nurse. Currently, cerebral pathologies are rarely screened routinely, and the brain is seldom monitored in hospital settings. VEEGix makes monitoring brain activity as straightforward as checking blood pressure.”
Jérôme Arnaud, COO of NeuroServo, added, “VEEGix is a revolutionary EEG platform offering unprecedented accuracy.
This FDA clearance is just the beginning. We are already preparing a submission for a new version of VEEGix, featuring additional electrodes and incorporating Artificial Intelligence algorithms to assist intensivists in diagnosing non-convulsive seizures. Other pathologies are coming next.”
About NeuroServo
NeuroServo is committed to advancing cerebral monitoring technology, making it accessible and straightforward for healthcare professionals to use in critical care settings. By integrating innovative hardware with AI-driven analysis, NeuroServo aims to revolutionize how brain health is assessed and managed in real-time.
NeuroServo’s development and growth have been supported by prominent financial investors, including Desjardins Capital, Investissement Québec, CABHI, C2MI, and other private investors.
JEROME ARNAUD
NeuroServo
+1 514-536-0250
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.