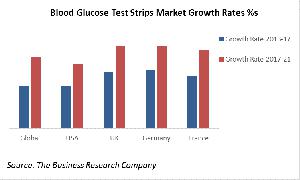
Public Policy Restrains Blood Glucose Test Strips Market Growth
LONDON, GREATER LONDON, UK, June 7, 2018 /EINPresswire.com/ -- The market for the blood glucose test strips used by diabetes patients to monitor their blood glucose levels is significantly affected by government policies, a new report from The Business Research Company shows. Many countries aim to encourage healthy lifestyles to reduce diabetes 2 prevalence levels, to regulate blood test strips quality and to curb government expenditure on items such as blood glucose test strips.
Governments are giving special attention to trying to curb the rising prevalence of diabetes. Public programs and initiatives are aimed at promoting healthy lifestyles and increasing awareness of the risk of developing diabetes. In the USA, for example, government agencies and health departments are aiming at improving the lifestyle of people who are at a higher risk of developing type II diabetes through the National Diabetes Prevention Program, which is a public-private partnership of community organizations, private insurers, and health care organizations. In Germany, the German Centre for Diabetes Research (DZD) seeks to develop novel strategies for successfully detecting, preventing and treating diabetes.
As the chart suggests, government efforts to rein in the diabetes 'epidemic' are expected to have limited success: in countries which are undertaking these initiatives growth of the sales of the blood glucose test strips that are used to monitor blood glucose levels in patients are predicted to grow nearly as fast (USA) or faster (Germany) than the global average.
Lower Reimbursements For Test Strips
Government reimbursements for blood glucose test strips have been reduced in many countries in a move to reduce the healthcare burden. For example, Medicare in the USA and the UK’s National Health Service reduced their coverage for the strips and even restricted patients' strip volume entitlements. Co-payments by patients for the test strips have increased, leading to higher expenditure for diabetic patients, indirectly affecting the consumption of test strips, which is nevertheless predicted to grow strongly. France, however, has taken a different line. In May 2017 the French Health Ministry granted national reimbursement across France for FreeStyle Libre – Abbott’s blood glucose monitoring technology. This reimbursement decision is expected to help both people with Type I diabetes who may use insulin and need to test multiple times per day and those with Type II diabetes. Reimbursement for test strips in most Asia-Pacific countries is limited; in China, for instance, there is no public reimbursement scheme covering diabetes test strips. Exceptions here are Australia and Japan which have generous systems for reimbursement and diabetes management.
Stricter Regulations
Regulatory changes are a further aspect of public policy that impacts the blood glucose test strip market. For instance, in 2014 the USA's Federal Drugs Administration issued new guidelines for blood glucose test strips and meters. The guidelines included provisions specific to generic test strips that are usuallyn less-expensive but have not been subject to high requirements for accuracy. The new FDA rules say these manufacturers should "ensure that they are aware of any design changes to the meter [used with the strip] because such changes could affect compatibility of the test strip with the meter." The FDA is also reviewing the manufacturers “test strip lot release” criteria, which was earlier not part of the review process for clearance. This change is expected to ensure standard levels of performance of strips in the market, but may result in higher prices for the generic test strips. Because the FDA is highly influential, other regulators, such as that of the European Union, may introduce similar restrictions.
Where To Learn More
Read the Blood Glucose Test Strip Market Global Report 2018 from The Business Research Company for information on the following:
Markets Covered: Blood glucose test strip market, blood glucose strips using glucose dehydrogenase, blood glucose strip using glucose oxidase, thick film, thin film and optical blood glucose strips
Blood Glucose Test Strip Companies Covered: Roche, Johnson & Johnson, Ascensia, Abbott, Nipro Diagnostics, Terumo, Arkray, Omron
Regions: North America, Asia Pacific, Western Europe, South America, Eastern Europe, Middle East, Africa.
Countries: USA, China, Japan, Germany, Brazil, France, Italy, UK, Australia, India, Spain, Russia.
Time Series: Five years historic (2013-17) and forecast (2017-21).
Data: Blood glucose test strip market size and growth for 7 regions and 12 countries; global regional and country blood glucose test strip market size and growth segmented by type of technology and by electrode material; global, regional and country blood glucose test strips per capita consumption and market size as a percentage of GDP 2013-21; Roche, Johnson & Johnson, Ascensia, Abbott, Nipro Diagnostics, Terumo, Arkray, Omron financial performance 2013-21
Other Information: PESTEL analysis, drivers and restraints, customer and operational insights, blood glucose test strip market by region and country covering opportunities, associations, investment and expansion plans, corporate tax structure and competitive landscape; blood glucose test strip market market trends and strategies.
Sourcing and Referencing: Data and analysis throughout the report are sourced using end notes. Sources include primary as well as extensive secondary research.
Related Reports: Pharmaceutical Market Global Report 2018
Biologics Market Global Report 2018
Oliver Guirdham
The Business Research Company
+44 207 1930 708
email us here